美国Ligand PeliCRM197是一种使用Pelican Expression Technology系统在荧光假单胞菌中表达的重组形式。PeliCRM197在结构和功能上与白喉棒状杆菌产生的突变蛋白相同,可提供满足各临床研究或商业化生产要求的研究等级或者cGMP等级的规格。

品牌列表
-
仪器设备

-
试剂耗材

- MineBio 精选
- 人AB血清(Human AB Serum)
- Akron CGT cGMP辅助材料
- Atelerix 常温保存凝胶
- BioLamina人类重组层粘连蛋白
- Cellagen Technology 小分子抑制剂
- Ethos (Exocell) 肾病学检测试剂盒
- GA international 实验室标签解决方案
- InSphero 3D细胞培养耗材
- 嘉士腾 类器官培养试剂
- Ligand PeliCRM197 白喉毒素突变体
- NETRI 神经芯片
- Nordmark 胶原酶
- NIBSC 标准品
- Polysciences转染试剂和化学品
- QKine 细胞因子
- Revvity LentiBOOST慢病毒转导增强剂
- RoosterBio 干细胞培养整体解决方案
- Sexton 人血小板裂解液
- Solallis 化学成分限定的无血清培养基
- Visual Protein 免疫印迹相关试剂
-
细胞

-
生物样本库

-
产品名称品牌货号规格询价详情备注
CRM197产品介绍
CRM197是一种将52位的甘氨酸替换为谷氨酸的单一突变的无毒性的白喉毒素突变体(文献1,2),致使白喉毒素酶活性位点发生改变,从而不能对细胞产生毒性作用,但在抗原性和免疫原性上仍和天然白喉毒素保持一致。CRM197 白喉毒素突变体是一种研究明确的蛋白质,作为多糖和半抗原的载体使其具有免疫原性。它被用作一种载体蛋白,用于许多已经被批准的疫苗,比如脑膜炎球菌、乙型流感嗜血杆菌和肺炎球菌感染的结合疫苗。
PeliCRM CRM197 白喉毒素突变体是一种使用Pelican Expression Technology系统在荧光假单胞菌中表达的重组形式。CRM197 白喉毒素突变体是结合疫苗的理想载体蛋白或免疫佐剂。通过物理化学特性和体内研究表明,PeliCRM197 白喉毒素突变体在结构和功能上与白喉棒状杆菌产生的突变蛋白相同。目前的研究和开发领域包括免疫疗法和抗成瘾性疫苗。可提供满足任何临床研究或商业化生产要求的研究等级或者cGMP等级的规格。自1990年以来,CRM197 白喉毒素突变体已被商业应用于预防性疫苗中。
2020年Pfenex被Ligand Pharmaceuticals收购,现在CRM197也被称为PeliCRM197。

CRM197产品特点
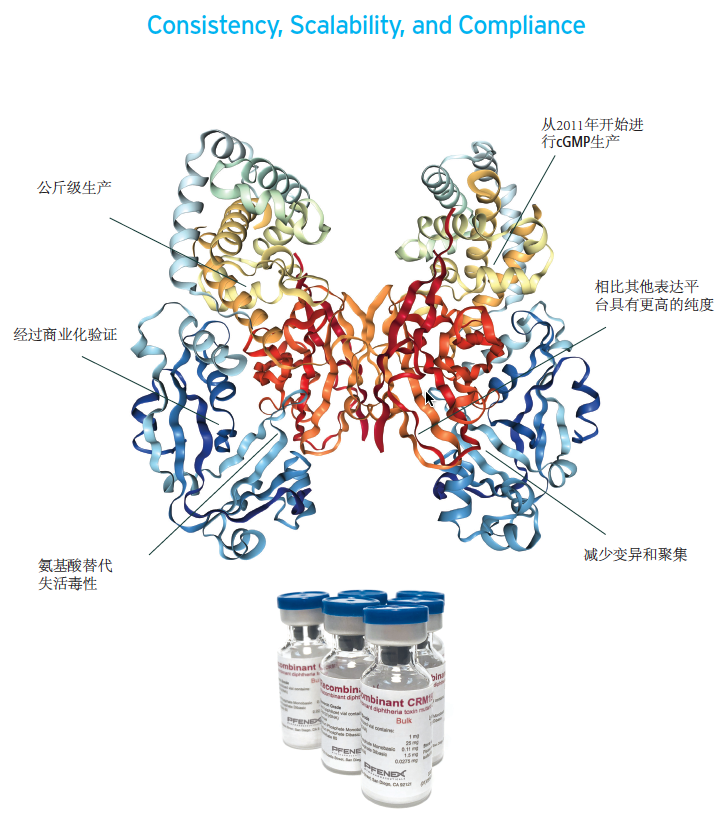
· 避免了在病原微生物中缓慢,复杂和昂贵的生产
· 与其他的表达系统获得的CRM197相比,PeliCRM197纯度更高
· 提供研究用和cGMP级的重组PeliCRM197载体蛋白(3)
· 从研究等级(mg级)到cGMP级(kg级)的产品和工艺保持一致性
· 加速疫苗研发进入临床,是疫苗开发和商业化的经济有效的关键材料来源
· 可提供生物制品生产主文件(Master File),用于支持FDA临床监管申报和美国以外区域的同等机构申报
· 生产工艺经过针对Pneumosil疫苗开发的WHO预审
注:(3)重组CRM197(bulk)产品是根据现行cGMP生产规范和ICH Q7和ICH Q11(视情况而定)指导原则制造的,重组CRM197(bulk)作为活性物质起始材料,用于最终用户进一步制造合适的原料药和/或药品。
CRM197使用方法
1、收到PeliCRM197冻干粉后请保存在2-8°C。
2、使用前,用无菌去离子水将冻干的PeliCRM197无菌复原至4 mg/mL的浓度。
3、配置好的重组PeliCRM197应在2-8°C下储存。
产品参数
来源: 荧光假单胞菌重组表


 MineBio 精选
MineBio 精选  BioLamina人类重组层粘连蛋白
BioLamina人类重组层粘连蛋白 













